The Flower That Runs the World’s Most Dangerous Pharmacy
How a roadside weed became the original heart drug — and is now turning up in cancer laboratories with a completely different job description
Eswar Krishnan, MD
April 10, 2026 · 14 min read
There’s a plant growing in English hedgerows and cottage gardens right now that contains one of the most medically useful — and most dangerous — chemicals in the history of pharmacy. Gardeners grow it for its tall spires of purple bells. Bees love it. Children’s books feature it. And for about 240 years, doctors have been carefully, nervously, extracting its chemistry to keep failing hearts alive.
The foxglove. Digitalis purpurea. Unremarkable to look at. Extraordinary to understand.
What follows isn’t a wellness post. It’s a story about a plant that sits at an almost uncomfortably thin line between medicine and poison — and how researchers in 2025 and 2026 are discovering it might have a completely different career ahead of it, one involving your immune system and some of the hardest-to-treat cancers we know.
How a Birmingham Doctor Changed Everything
The year is 1775. A physician named William Withering is working in Birmingham, England, and one of his patients — a woman with severe edema, the kind where fluid fills the body cavities and the legs swell grotesquely — has run out of options. The standard treatments aren’t working. Withering expects her to die.
Then he discovers she’s recovered. Not through his ministrations. She’d been quietly taking a herbal concoction from a local woman in Shropshire. Withering, who happened to be a gifted botanist as well as a physician, looked at the mixture of twenty-odd herbs and immediately recognized the likely active ingredient.
Foxglove.
He spent the next ten years methodically documenting every case he treated with it. Not cherry-picking the successes — a level of scientific honesty remarkable for his era. He included failures, toxicities, and patients who died. In 1785, he published An Account of the Foxglove and Some of Its Medical Uses, describing 158 patients. Of those, 101 with congestive heart failure got better. His dosing calculations were so precise that modern analysis puts them only slightly below what we use today.
“Truth and Science would condemn the procedure. I have therefore mentioned every case... proper or improper, successful or otherwise.”
— William Withering, 1785
That book is one of the most important documents in pharmacological history. Withering knew it. His portrait — the only one from life — shows him holding a foxglove sprig. His epitaph at Edgbaston Old Church is carved with the plant. The man and the flower became inseparable.
What he didn’t know, and what took another century to understand, was whyit worked.
The Chemistry of the Foxglove
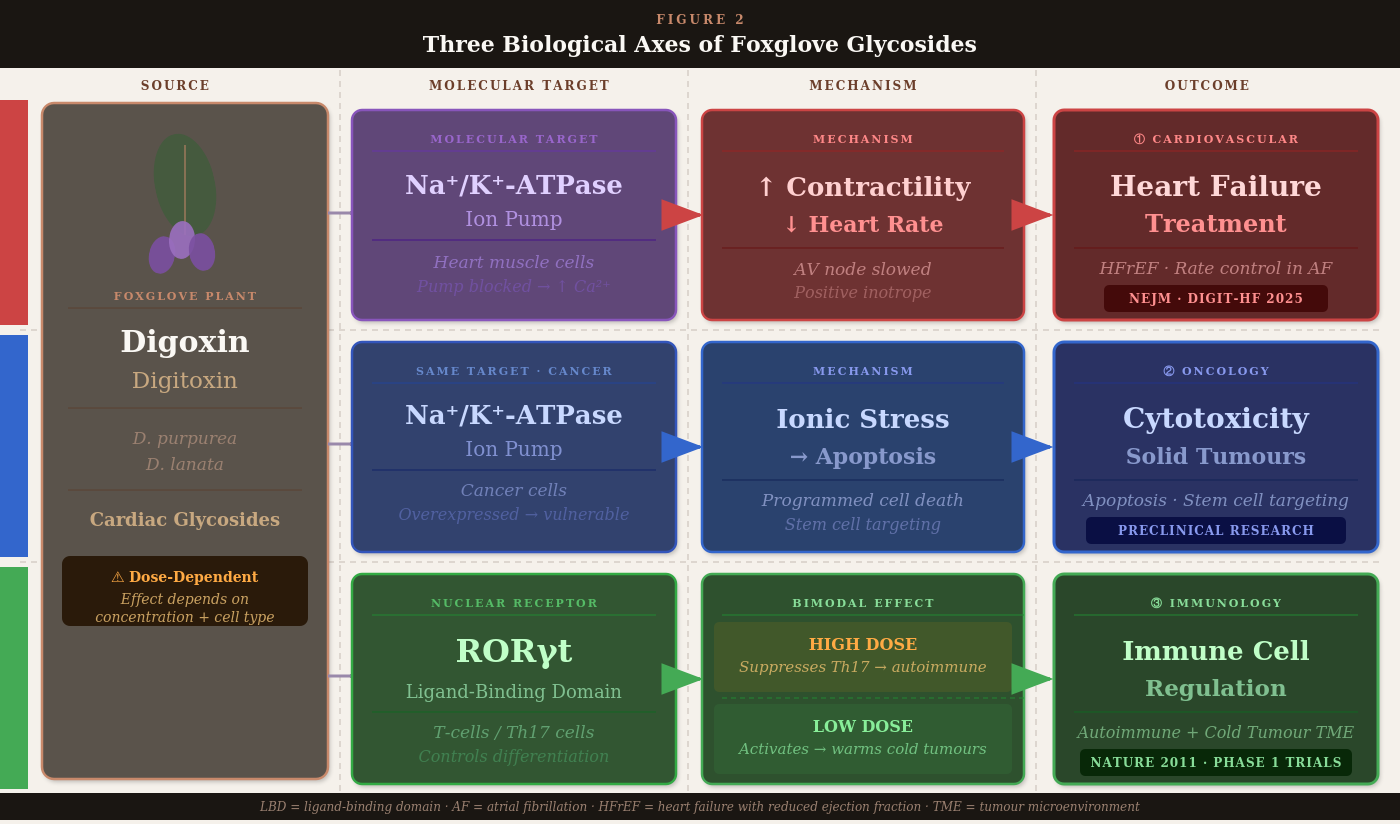
The foxglove produces a class of chemicals called cardiac glycosides — the name a clue to their structure: a sugar molecule attached to a steroid-like core. The plant makes them presumably as a defense against insects. For mammals with hearts, these compounds do something very specific and very powerful.
They block a molecular pump called Na⁺/K⁺-ATPase — an enzyme embedded in every heart muscle cell that normally shuffles sodium out and potassium in. When you block this pump, sodium accumulates inside the cell, causing calcium to flood in. More calcium means stronger, more forceful contractions.
In a weakening heart, that’s the difference between barely pumping and actually pumping.
The main pharmaceutical compounds extracted from foxglove are three siblings with distinct personalities:
Digoxin (from Digitalis lanata, the woolly foxglove): The one most doctors and patients have heard of. A “positive inotrope” — it makes the heart beat harder. It also slows the heart rate by affecting the electrical conduction system, which makes it useful for atrial fibrillation: the fast, chaotic rhythm that affects millions of people.
Digitoxin (from Digitalis purpurea, the common purple foxglove): The older sibling. Similar mechanism, longer-acting, and critically — cleared by the liver rather than the kidneys. This matters more than it sounds, as we’ll get to.
Lanatoside C: A faster-acting glycoside used in acute situations where you need to stabilize a rhythm quickly and can’t wait for slower-clearing agents to take effect.
FIGURE 1 — PLACEHOLDER
Purple foxglove (D. purpurea) vs. Woolly foxglove (D. lanata): leaf morphology comparison, distribution of active glycoside content by plant part, and primary commercial growing regions for pharmaceutical extraction
The Narrow Window
Here’s what makes foxglove so peculiar as a medicine. Its therapeutic window — the gap between “helpful dose” and “harmful dose” — is one of the narrowest in all of pharmacology. The dose that strengthens a failing heart sits uncomfortably close to the dose that causes fatal arrhythmias.
THE NUMBERS
The blood level of digoxin that helps a heart failure patient sits around 0.5–0.9 nanograms per milliliter. Toxicity begins at roughly 2 nanograms per milliliter. For context: a human hair is about 70,000 nanograms. We’re managing people on differences that are invisible to the naked eye.
Withering noticed this in 1785. He described toxicity symptoms with clinical clarity that would be recognized by any emergency physician today: irregular pulse, visual disturbances including a famous yellow-tint to vision, nausea, and ultimately fatal rhythm disturbances. The Victorians called it “foxglove sickness.” Modern medicine calls it digitalis toxicity, and it still kills people who are overdosed or whose kidney function changes unexpectedly.
This toxicity is why, over the last few decades, digoxin prescriptions declined sharply. Newer drugs — beta-blockers, ACE inhibitors, SGLT2 inhibitors — arrived with better safety profiles and more predictable behavior. Digoxin was increasingly treated as a relic of an earlier era.
Then, in August 2025, a clinical trial made a lot of cardiologists look up from what they were doing.
The DIGIT-HF Trial: Digitoxin’s Comeback
The DIGIT-HF trial — a double-blind, placebo-controlled study from 55 sites across Germany, Austria, and Serbia — enrolled 1,240 patients with advanced heart failure and reduced ejection fraction. These were sick patients: most were in NYHA class III (symptomatic at minimal exertion), and all were already on contemporary guideline-directed therapies including the newer drugs that have transformed heart failure treatment.
The question was whether adding digitoxin — not digoxin, but its liver-cleared cousin — would make any additional difference.
Published in the New England Journal of Medicine in August 2025, the answer was yes. Digitoxin reduced the combined risk of death from any cause or hospitalization for worsening heart failure by an absolute 4.6 percentage points over a median follow-up of three years.
The effect size is modest. But what makes it interesting is the population and the mechanism. The benefit held across patients taking modern quadruple therapy — the most aggressive current treatment protocol.
And the lead investigator specifically called out that digitoxin may be particularly useful for patients with impaired kidney function: precisely the population that struggles most with digoxin’s renal clearance requirements.
Because digitoxin is cleared by the liver, its blood concentrations remain stable even as kidney function deteriorates. No complex dose adjustments every time creatinine creeps up. No panic when a heart failure patient’s kidneys — which often go hand-in-hand with the failing heart — start to worsen. For clinicians managing this very common and very difficult combination, that’s a practically meaningful difference.
The Part Nobody Expected: Cancer
Here is where the foxglove’s story takes a turn that would have genuinely astonished William Withering.
The same Na⁺/K⁺-ATPase pump that foxglove glycosides inhibit in heart muscle cells is overexpressed in multiple cancer cell types. When you block it in a cancer cell, the consequences are considerably more lethal than in a normal cell. Cancer cells, with their high metabolic demands and already-stressed physiology, are more vulnerable to the disruption of ionic balance.
Laboratory studies have shown that digitalis-derived compounds are highly cytotoxic — cell-killing — against a range of human cancer cell lines. The mechanism involves triggering apoptosis: the cell’s own programmed self-destruction pathway, which cancer cells have typically evolved to evade. Forcing that pathway back on is something oncologists have been trying to do with many different compounds.
More intriguing is research targeting cancer stem cells — the small subpopulation of cells within a tumor that are most resistant to conventional chemotherapy, and that are thought responsible for relapse and metastasis after treatment appears successful. Early research suggests that cardiac glycosides may have activity against this population that standard chemotherapy misses entirely. The mechanism isn’t fully established, but it involves the same pump inhibition affecting stem cell signaling pathways.
This is preclinical data. It would be dishonest to call it a cancer treatment. But it’s enough that multiple research groups are pursuing it — and it intersects with a much larger and stranger story about the immune system.
RORγt: The Molecular Switch Nobody Knew Was There
In 2006, immunologists identified a transcription factor — a molecular switch that controls gene expression — called RORγt (pronounced “ROR-gamma-t”). It turned out to be the master regulator of a class of immune cells called Th17 cells.
Th17 cells produce a signaling molecule called IL-17. When these cells are overactive, they drive inflammatory and autoimmune conditions: rheumatoid arthritis, psoriasis, inflammatory bowel disease. Block RORγt, the logic went, and you suppress Th17-driven inflammation. Pharmaceutical companies began racing to find oral drugs that could achieve what expensive injectable biologics were already doing.
2006
RORγt identified as the master regulator of Th17 cell differentiation — immediately flagged as a high-value drug target for autoimmune disease
2011
A Nature paper reports that digoxin — the foxglove drug — binds to the RORγt ligand-binding domain and blocks Th17 differentiation. A completely separate mechanism from anything happening in the heart
2012–2017
Major pharmaceutical investment in RORγt inhibitors. Clinical trials run against autoimmune indications. Results are underwhelming versus biologics; safety concerns emerge around thymic T-cell effects
2018–2023
The inhibitor program cools off. But separately, researchers begin looking at what happens when you go the other direction — activating RORγt rather than suppressing it
2024–2026
The oncology pivot: RORγt agonists enter early clinical trials to “heat up” cold tumors and make them visible to checkpoint immunotherapy. Digoxin’s role as the molecule that first mapped this receptor comes back into focus
The Flip: From Suppressor to Activator
Researchers had been thinking about RORγt as something to switch off — to dampen overactive inflammation. The oncology pivot asked a different question: what if you switched it on?
In cancer, a well-documented problem is the cold tumor: a tumor that has managed to exclude immune cells from its microenvironment, making itself essentially invisible to the body’s defenses. Checkpoint inhibitors — the cancer drugs that have transformed oncology over the last decade — work best in tumors already infiltrated by immune cells. Cold tumors don’t respond nearly as well. And many common cancers run cold.
RORγt agonists — molecules that activate rather than block the receptor — appear capable of recruiting immune effector cells into cold tumor environments. The Th17 pathway, when activated appropriately in a tumor context, can drive an immune infiltration that checkpoint inhibitors then amplify. You warm the tumor up first, then send in the blockers.
This is not science fiction. A first-in-class synthetic RORγt agonist — cintirorgon (LYC-55716) — completed a Phase 1 trial in patients with relapsed or refractory metastatic cancers. No dose-limiting toxicities occurred among the 32 enrolled patients across a range of doses, and early antitumor signals were observed.
The foxglove connection here is structural and mechanistic rather than direct. Digoxin and its derivatives were first used to map the RORγt binding site and to demonstrate its functional importance. The synthetic agonist programs that followed built on that knowledge. Whether digitalis-derived molecules themselves will be developed as RORγt agonists remains an open question — the margin between their cardiac effects and their immunological effects requires careful pharmacological navigation — but the research is active.
The Dose Makes the Poison — and the Medicine
What connects all of these threads is a concept that goes back to the 16th-century physician Paracelsus: sola dosis facit venenum. The dose alone makes the poison.
Foxglove at high concentrations: cardiac toxicity, fatal arrhythmias, death.
At therapeutic concentrations in a failing heart: life-saving inotropy and rate control.
At lower concentrations affecting RORγt: immune modulation with potential autoimmune and oncologic applications.
At concentrations affecting cancer cell pumps: potential cytotoxicity against tumor cells that overexpress the target.
The same molecule. Different doses. Wildly different biological conversations.
This is a recurring pattern in pharmacology, but foxglove makes it almost embarrassingly legible. It’s a chemical with so many conversations going on with the human body that we are still — 240 years after Withering’s book — finding new ones.
The Flavonoids — The Part Nobody Talks About
The cardiac glycosides get all the attention, but foxglove leaves contain other compounds that researchers are only now beginning to characterize seriously.
One is scutellarein, a flavonoid with antioxidant properties entirely separate from any cardiac effect. Early research suggests anti-inflammatory activity through mechanisms distinct from the glycoside pathway. It’s far too early to say where this leads, but it illustrates a broader point: plants are not single-compound factories. The foxglove leaf is a complex biochemistry experiment, and we’ve mostly been studying one product line.
Traditional medicine systems — particularly in parts of India — have used topical preparations of digitalis glycosides in ointments for severe burns, where the compounds are thought to stimulate local circulation and aid tissue healing. This sits outside mainstream Western pharmacology and hasn’t been rigorously studied in clinical trials. But it’s not implausible given what we understand about vascular effects. It’s a loose thread, and loose threads sometimes lead somewhere.
What We Know, What We Don’t
The honest position on foxglove in 2026:
Well-established: Digoxin and digitoxin are real medicines with real cardiovascular applications. The DIGIT-HF trial provides solid evidence that digitoxin reduces hospitalization and death in certain heart failure patients, particularly those with renal impairment. Both carry a genuinely narrow therapeutic window requiring careful clinical management.
Promising but not proven: Anticancer applications of cardiac glycosides are an active and intriguing area of lab research. The findings are consistent across multiple cell line studies. Clinical validation in humans is limited.
Scientifically coherent but speculative: The path from “digoxin binds RORγt” to “foxglove-derived molecules treat cold tumors” runs through a lot of unsolved pharmacological and safety problems. The synthetic agonist programs inspired by this discovery are further along than the digitalis-derived work itself.
Withering’s own instinct, looking at his 1785 data, seems right for 2026 as well: document everything, claim only what the evidence supports, and leave room for what comes next.
The foxglove has been growing in the hedgerows for millions of years. Its chemistry has been tested in folk medicine for centuries, in systematic clinical trials for 240 years, and in molecular biology laboratories for the last few decades. Every time we think we understand what it does, it shows us another room.x
That’s either a sign of a very strange plant, or a sign of how much we still have to learn about the cells we’re made of.
VERY IMPORTANT: This post is written for general interest and educational purposes. None of the content above constitutes medical advice. Digitalis-derived medications are prescription drugs with serious and even fatal toxicity potential and should never be self-administered or sourced from the plant directly.
SOURCES & FURTHER READING
Bavendiek U et al. “Digitoxin in Patients with Heart Failure and Reduced Ejection Fraction.” N Engl J Med2025;393(12):1155–1165.
Karaś K et al. “Digoxin, an Overlooked Agonist of RORγ/RORγT.” Frontiers in Pharmacology 2018;9:1460.
Withering W. An Account of the Foxglove and Some of Its Medical Uses. Birmingham: M. Swinney, 1785.
Mahalingam D et al. “Phase 1 Open-Label, Multicenter Study of First-in-Class RORγ Agonist LYC-55716 (Cintirorgon).” Clin Cancer Res 2019;25(12):3508–3516.
“Digitalis – from Withering to the 21st century.” British Journal of Cardiology, August 2024.
Nature Reviews Cardiology. “Benefit of digitoxin therapy for HFrEF.” Vol. 22, p. 842, September 2025.